Monocyte
Inflammation Assays
- Inflammasome/LPS cytokine release
- Predictive immunotox CRA
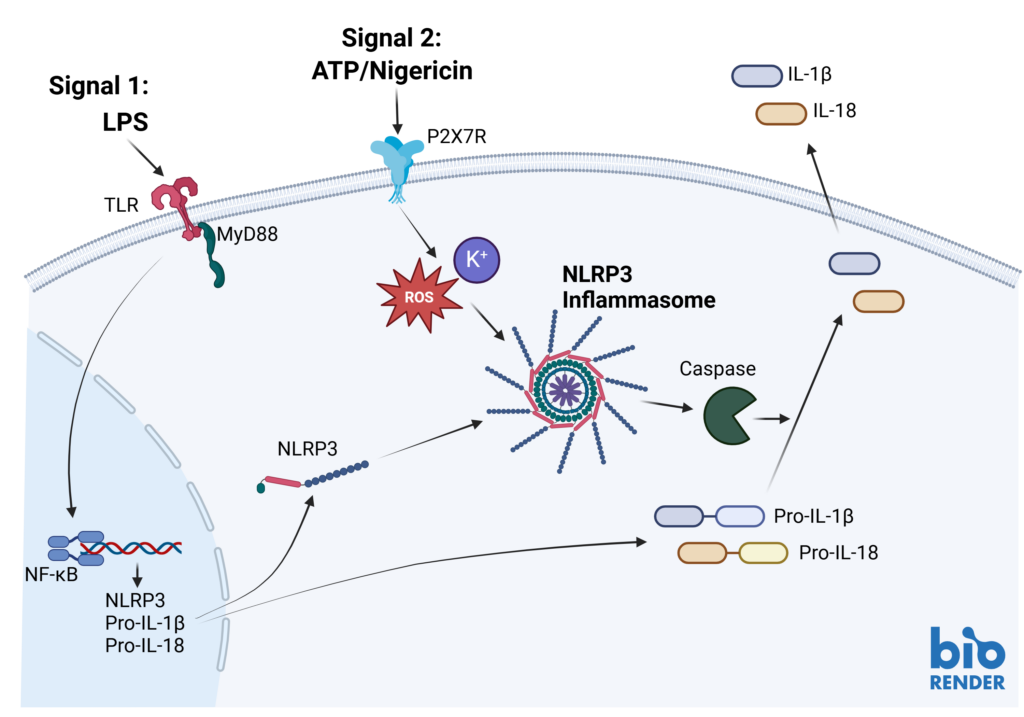
Inflammasomes are key signaling platforms within the immune system. Inflammasome complexes form in response to infection, tissue damage or metabolic imbalances. Once formed the inflammasomes activate Caspase 1 which in turn activated the pro-inflammatory cytokines IL-1β and IL-18. Targeting the inflammasome and the resulting signaling pathways is a useful tool in modulating the immune system.


Effect of MCC950 on Inflammasome activation – IL-1β and IL-18.
PBMC from three healthy donors were pretreated with MCC950, vehicle (DMSO) or media for 1 hours prior to stimulation with LPS for 4 hours. Nigericin was then added for a further 45 minutes prior to harvest of the cell culture supernatant for quantification of (A) IL-1β and (B) IL-18 by TR-FRET and ELISA respectively. Data points show mean +/- SEM of technical replicates. One-way ANOVA with Dunnett’s multiple comparisons test comparing stimulated (media) to unstimulated, LPS alone, vehicle and MCC950 to vehicle; *<0.05, **<0.01, ***<0.001.
Other formats: PBMC, macrophage subsets, microglia
Cytokine release assay can be used to “de-risk” any potential unwanted effects of novel biotherapeutics in accordance with FDA and EMA recommended guidelines.
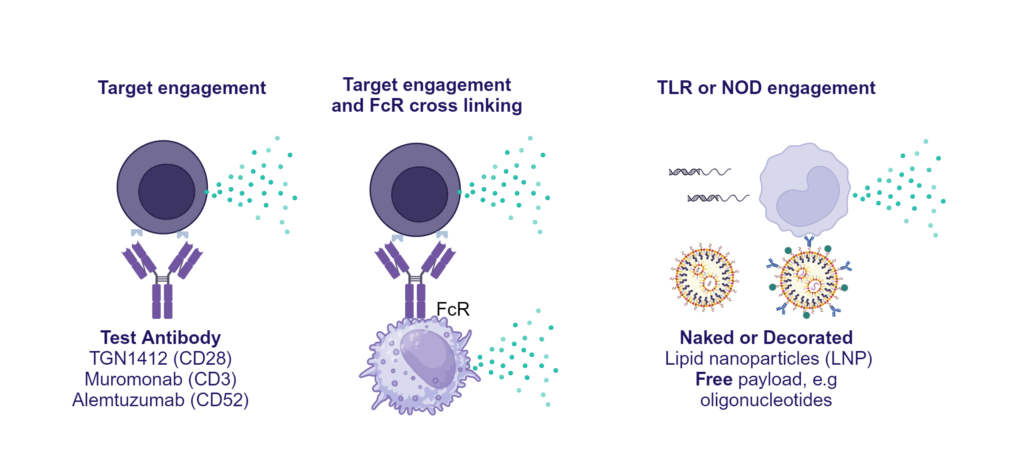
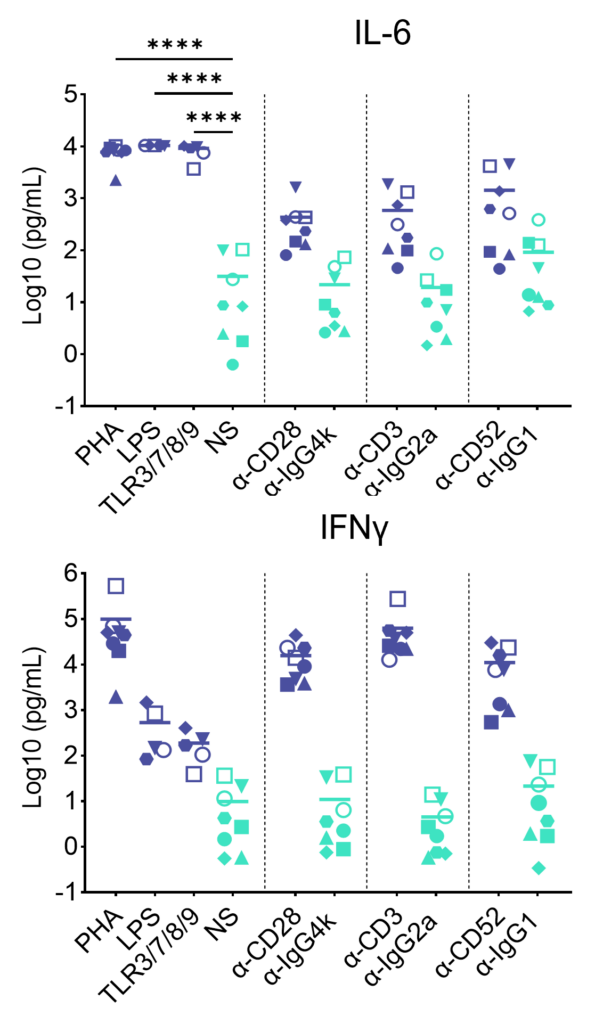

Preclinical safety cytokine release assay (CRA)
PBMC were added to wells coated with Alemtuzumab, Muromonab, TGN1412 or with isotype control antibodies, to induce cytokine release, or were stimulated with PHA, LPS, or a cocktail of TLR3/7/8/9 agonists. Cytokine release was measured at 48hr post stimulation. NS = Not stimulated. Data shown is mean of a minimum of five donors. Statistics show one way ANOVA with Tukey’s multiple comparisons test. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
Other readouts: IL-8, IL-13, IL-4, IL-12p70, IL-10, IL-1β
Other formats: Whole Blood CRA
Other immuno-tox assays: include ADCC/ CDC/ ADCP

Drug Discovery Tool
Find the right immune assay for your therapeutic area, modality and target using this interactive Drug Discovery Tool.
